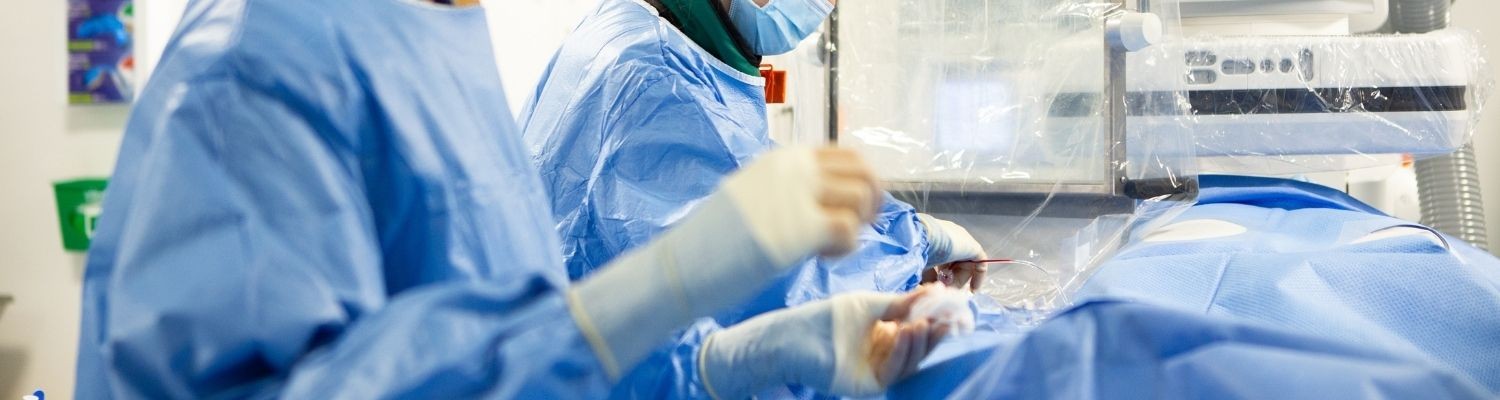
The intensive care unit (ICU), also known as critical care or intensive treatment unit (ITU), is where we monitor and care for patients who are critically ill. These patients often have a problem with one or more vital organs.
Patients may be admitted to ICU if they have had planned or emergency major surgery, have serious short-term conditions such as a stroke, or a serious infection like sepsis or severe pneumonia.
Our ICUs have 24-hour cover by professionals specially trained in critical care medicine. Consultants are supported by a multidisciplinary team, who work together to care for our patients.
Royal Free Hospital ICU
The ICU at the Royal Free Hospital has 50 beds between the 3rd and 4th floors of the hospital and admits over 2,000 patients each year.
The hospital provides monitoring and critical care for local patients and patients referred from hospitals around the UK who come for our specialist services.
Patients may be referred to our hospital for liver failure, complex liver surgery, heart attacks or heart failure, kidney failure, complex neurological needs, or infectious diseases.
Barnet Hospital ICU
Barnet Hospital’s ICU has 18 beds in two adjacent units: ICU North and ICU South. There are five beds in side rooms and the ICU admits around 800 patients per year.
Patients from different specialties, including general acute medical patients, emergency and elective surgery, and acute renal replacement therapies, all receive treatment in Barnet ICU.
Patients receive a weekly multidisciplinary clinical review.
If you are a medical professional making a referral to ICU, please contact the Royal Free Hospital switchboard on 020 7794 0500 and ask for the ICU consultant on call. You will be asked for information on the following documents when making a referral:
Please have as much information ready as possible.
We work with ACCESS London to help patients move between hospitals safely.
Patient outcomes are reported to the Intensive Care National Audit & Research Centre (ICNARC).
The ICU is an unfamiliar environment for most people and can feel very different to a normal ward.
When someone is first admitted to ICU, visitors will usually be asked to wait in our reception area for a brief time. This is so the team can assess the patient, take any samples or scans needed, and begin treatment where possible. This is usually a very busy period, and we thank you for your patience. We will aim to keep you updated as much as possible.
Being critically unwell in ICU will cause physical changes to the patient’s body and may have a significant emotional and psychological impact on the patient and their family. It is normal to feel frightened, worried, and confused. We are here to provide any help you may need.
Our visiting hours are between 12 noon and 7.30pm, and a maximum of two visitors are allowed at the patient’s bedside at any one time. However, there may be circumstances when these details are changed. The decision to do this is taken very seriously by senior doctors and nurses, to prioritise patient and visitor care and safety.
It is a good idea to check visiting hours and bedside capacity with the nurse looking after your patient before you arrive by phone.
Please ensure you clean your hands when arriving and leaving ICU, and avoid visiting if you are ill, to reduce spreading infection.
An ICU can be an overwhelming place for patients and their loved ones. You can find more information on what you can expect when visiting an ICU on the NHS website.
Information sharing
It is helpful to identify a next of kin and one or two other contacts who can be given updates about the patient and update the wider social circle as they wish.
All patients have a right to confidentiality. Our staff have an ethical and legal obligation to ensure confidentiality of patient information, and in some circumstances will not be able to give out detailed information.
To help with this, patients and visitors often create a password that can be given by a family member to staff to confirm they are allowed to be given clinical updates and/or visit. This can help minimise confusion and make sure information is only shared with the correct people.
Please let the bedside nurse know if you would like to use a password and what the password is. They will record this in the patient’s notes and hand it over at every shift.
Below are some ideas on ways to be involved in the patient’s care and recovery, but you are under no pressure to do any of these. Please talk to a member of staff if you would like any guidance.
Motivation boards
For patients who have been with us for some time, we often find it is helpful to work with families to create a board for them to look at.
The board might include simple reorientation information, such as the date, time and place, or photos of family or pets.
It could record the patient’s likes and dislikes, hobbies, favourite songs, TV shows or any topics the patient is interested in.
Hospital diary
For many patients, their memories of ICU can be confused and incomplete. It can be useful to keep a diary for them to look through when they have left ICU and are feeling better.
The diary might include changes or progress in their care and treatment, and news about what has been happening at home.
Understanding what we do in ICU
Read through the information on this website. Often patients find it comforting to hear some of the explanations around what is happening from someone they know and trust.
Our ICU videos will tell you more about the team, the work we do and how we support patients through their illness.
Look after yourself and each other
Having someone you care about being critically ill in ICU is a very stressful experience, with potential emotional traumas, that can take focus away from keeping yourself healthy. We take this care and support very seriously.
Make sure to eat and take fluids regularly. Your GP may be able to support you with taking time off work or contacting psychological or local support services.
Our chaplaincy and spiritual care team also support patients and families of all beliefs, faiths and philosophy of life during their time in the ICU. You do not need to consider yourself religious to seek their support.
Recovery and moving to a ward
Patients recovering from being critically unwell will hopefully be discharged onto a ward.
This is a very positive step in recovery, however, it is natural for the patient to feel anxious about leaving ICU to go onto another ward. We understand this change can also cause anxiety for patients’ families.
We aim to ensure safe transition of care. When a patient is discharged from ICU, all decisions are carefully considered by the multidisciplinary team led by an ICU consultant.
The responsible medical or surgical team at the ICU will review the patient before they are discharged to the ward.
ICU nursing staff and therapists will communicate with staff on the ward about the patient’s care and ongoing plans for continuity of care. The patient at risk and resuscitation team (PARRT) will also visit patients to provide ongoing support to the patient and the ward team.
Patients will continue to be cared for by the multidisciplinary team. Being very unwell can have lasting effects, and recovery from an ICU stay can be a long journey. Patients may also need continuing support from community care teams after they leave hospital.
Psychological support
We have a dedicated ICU psychology service. Our psychologists support the needs of patients and their families during admission to ICU. They will help with assessing and managing a range of issues that can arise during admission, such as acute distress, trauma, cognitive difficulties, end of life and bereavement, behavioural problems impacting care or rehabilitation and more.
Their aim is to identify patients at risk of psychological distress after discharge and signpost them to appropriate services. Staff work directly with patients and loved ones during an ICU stay and beyond if needed.
Barnet Hospital follow-up clinic
At Barnet Hospital, we run a monthly follow-up clinic for patients who have been discharged from hospital after spending a long time in ICU. This clinic is run by ICU doctors, nurses, physiotherapists, occupational therapists, dieticians and psychologists.
This clinic helps patients who may face unique post-critical illness issues, with their recovery from clinical illness back to health.
Following the COVID-19 pandemic, over 130 patients who spent time in ICU attended this clinic.
Bereavement support
Sometimes, despite our best efforts, a patient will not survive their ICU stay. In this case, every effort will be made to ensure the patient is comfortable at the end of their life. We are here to support both patient and family throughout this process and have many experienced staff to assist us.
Our bereavement officers provide emotional and practical support to families and friends who have lost a loved one.
Their contact details can be found on the bereavement page of our website.
If you would like more information on organ donation, you may find the following resources useful.
NHS organ donation
Your feedback is important to us. If you have any concerns or questions, please first talk to a member of staff in the ICU or on the ward.
If your issue has not been resolved, or you would prefer to speak with someone outside of the department, please contact our Patient Advice and Liaison Service.
Introduction to our ICUs
This video provides information on: ICU staff members (00:29), why patients come to ICU (2:20), organ support (3:07), tubes and lines (4:14), medication (5:47).
Communications between patients and relatives/friends
This video provides information on: visiting ICU (00:00), video calls with relatives (00:53), motivational boards (1:03), communication with medical team (1:25), additional family support (2:10).
Recovery
This video provides information on: weaning from organ support (00:01), tracheostomy (00:35), emotional and psychological support (1:44), rehabilitation (2:30).
Research
This video provides information on research and recruitment into trials.
- advice and support on next of kin
- NHS advice on carers benefits
- chaplaincy services at the Royal Free London
- citizens advice: managing affairs for someone else
Information and support
- Healthtalk: a website with information about patient experiences of intensive care.
- HealthUnlocked intensive care forum: a forum for ICU patients and their loved ones.
- ICUsteps: a charity and support group for ICU patients and their loved ones that produces a detailed intensive care guide.
- Intensive Care Society: a professional body that provides information about intensive care for patients and relatives.
 Translate
Translate